SECOND LAW OF THERMODYNAMICS
Thermodynamics
An actual turbine does less work because of friction losses in the blades, leakage past the blades
and, to a lesser extent, mechanical friction. Turbine efficiency
, sometimes called isentropic
h t
turbine efficiency because an ideal turbine is defined as one which operates at constant entropy,
is defined as the ratio of the actual work done by the turbine W
t, actual
to the work that would be
done by the turbine if it were an ideal turbine Wt,ideal.
(1-26)
h t
Wt,actual
Wt,ideal
(1-27)
h
(hin
hout)actual
(hin
hout)ideal
where:
= turbine efficiency (no units)
h t
Wt,actual
= actual work done by the turbine (ft-lbf)
Wt,ideal
= work done by an ideal turbine (ft-lbf)
(hin - hout)actual
= actual enthalpy change of the working fluid (Btu/lbm)
(hin - hout)ideal
= actual enthalpy change of the working fluid in an ideal turbine
(Btu/lbm)
In
many
cases,
the
turbine
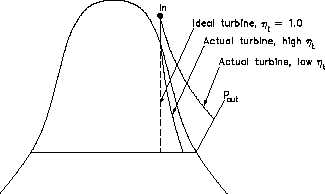
Figure 27 Comparison of Ideal and Actual Turbine Performances
efficiency
has been determined
h t
independently. This permits the
actual work done to be calculated
directly by multiplying the turbine
efficiency
by the work done by
h t
an ideal turbine under the same
conditions. For small turbines, the
turbine efficiency is generally 60%
to 80%; for large turbines, it is
generally about 90%.
The
actual
and
idealized
performances of a turbine may be
compared conveniently using a T-s
diagram. Figure 27 shows such a
comparison. The ideal case is a
constant entropy. It is represented
by a vertical line on the T-s diagram. The actual turbine involves an increase in entropy. The
smaller the increase in entropy, the closer the turbine efficiency
is to 1.0 or 100%.
h t
HT-01
Page 80
Rev. 0