Fundamentals of Chemistry
DOE-HDBK-1015/1-93
THE PERIODIC TABLE
Rev. 0
CH-01
Page 19
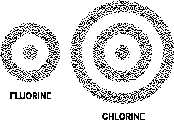
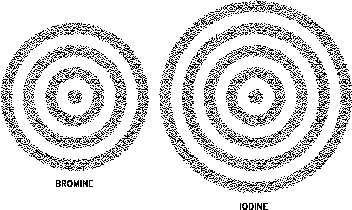
Figure 5 Electron Shells of Atoms
Chemical activity can also be determined from position in the periodic table. The most active
metals are the members of the Alkali Family, e.g., cesium (Cs) and francium (Fr). The most
active nonmetals are the members of the Halogen Family, e.g., fluorine (F) and chlorine (Cl).
The Noble Gases in Group 0 are inert. The activity of metals decreases when proceeding to the
right in the periodic table; the activity of nonmetals decreases when proceeding to the left.
Atomic Structure of Electrons
Based on experimental data, it is known that chemical reactions involve only the electrons in
atoms. In fact, only some of the electrons are involved. Because chemical properties are
periodic, there must also be a periodic characteristic about electrons. This characteristic is the
manner in which electrons are arranged in the atom. Electrons are in constant motion around
the nucleus. They have both kinetic and potential energy, and their total energy is the sum of
the two. The total energy is quantized; that is, there are definite, discrete values of total energy
that atomic electrons can possess. These energy states can be visualized as spherical shells
around the nucleus separated by forbidden areas where electrons cannot exist in a stable state.
This sort of arrangement is illustrated in Figure 5.
It is customary to speak of electron shells around the nucleus, and the shells are referred to by
number. The first, or No. 1, shell is the one nearest the nucleus; the second, or No. 2, shell is
next; then the third, or No. 3, shell; and so on in numerical order. In general, electrons closer
to the nucleus have a lower energy state. Atomic electrons always seek the lowest energy state
available.