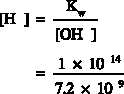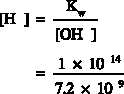Fundamentals of Chemistry
DOE-HDBK-1015/1-93
ACIDS, BASES, SALTS, AND pH
Rev. 0
CH-01
Page 51
Example:
The hydroxyl concentration [OH ] of a water solution at 25 C is
-
o
7.2 x 10 moles/liter. Calculate the pH of the solution.
-9
Solution:
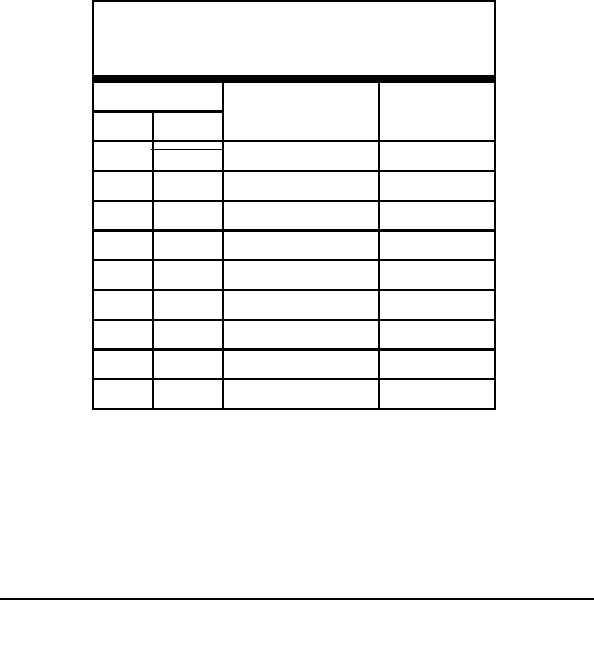
TABLE 5
Ion Product Constant and Neutral pH
for Water at Various Temperatures
Temperature
Ion Product Constant
pH of Pure
for Water
Water
F
C
64.4
18
0.64 x 10
7.10
-14
77
25
1.0 x 10
7.00
-14
140
60
8.9 x 10
6.54
-14
212
100
6.1 x 10
6.10
-13
302
150
2.2 x 10
5.83
-12
392
200
5.0 x 10
5.65
-12
482
250
6.6 x 10
5.59
-12
572
300
6.4 x 10
5.60
-12
662
350
4.7 x 10
5.66
-12
At 25?C, an equilibrium exists between pure molecular water and its ions. The [H ] equals the
+
[OH ] and both have values of 1 x 10 moles/liter. Using the pH definition, it follows that the
-
-7
pH of pure water at 25?C is 7. pH values less than 7 indicate an acidic solution and values
greater than 7 indicate a basic or alkaline solution.