Thermodynamics
TEMPERATURE AND PRESSURE MEASUREMENTS
Pressure
Pressure is a measure of the force exerted per unit area on the boundaries of a substance (or
system). It is caused by the collisions of the molecules of the substance with the boundaries of
the system. As molecules hit the walls, they exert forces that try to push the walls outward. The
forces resulting from all of these collisions cause the pressure exerted by a system on its
surroundings. Pressure is frequently measured in units of lbf/in2 (psi).
Pressure Scales
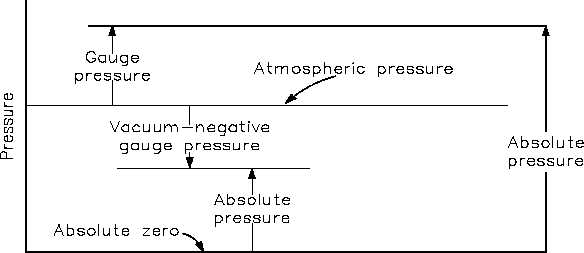
When pressure is measured relative to a perfect vacuum, it is called absolute pressure (psia);
when measured relative to atmospheric pressure (14.7 psi), it is called gauge pressure (psig). The
latter pressure scale was developed because almost all pressure gauges register zero when open
to the atmosphere. Therefore, pressure gauges measure the difference between the pressure of
the fluid to which they are connected and that of the surrounding air.
If the pressure is below that of the atmosphere, it is designated as a vacuum. A perfect vacuum
would correspond to absolute zero pressure. All values of absolute pressure are positive, because
a negative value would indicate tension, which is considered impossible in any fluid. Gauge
pressures are positive if they are above atmospheric pressure and negative if they are below
atmospheric pressure. Figure 2 shows the relationships between absolute, gauge, vacuum, and
atmospheric pressures, as do Equations 1-9 and 1-10.
Figure 2 Pressure Relationships
Rev. 0
Page 9
HT-01