Thermodynamics
SECOND LAW OF THERMODYNAMICS
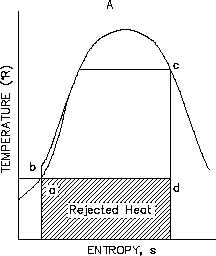
Figure 32 Rankine Cycle With Real v.s. Ideal
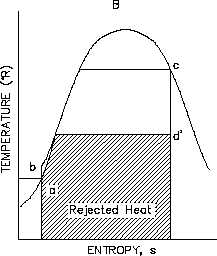
From the T-s diagram (Figure 32) it can also be seen that if an ideal component, in this case the
turbine, is replaced with a non-ideal component, the efficiency of the cycle will be reduced. This
is due to the fact that the non-ideal turbine incurs an increase in entropy which increases the area
under the T-s curve for the cycle. But the increase in the area of available energy (3-2-3’,
Figure 32) is less than the increase in area for unavailable energy (a-3-3’-b, Figure 32).
Figure 33 Rankine Cycle Efficiencies T-s
Rev. 0
Page 89
HT-01