Structure of Metals
DOE-HDBK-1017/1-93
POLMORPHISM
The alpha (a) phase is stable at room temperature and has a crystal system characterized
by three unequal axes at right angles.
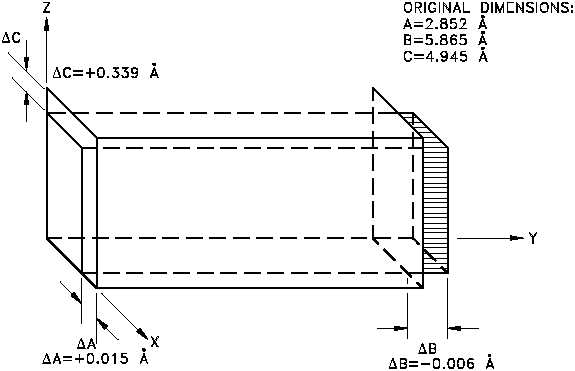
In the alpha phase, the properties of the lattice are different in the X, Y, and Z axes.
This is because of the regular recurring state of the atoms is different. Because of this
condition, when heated the phase expands in the X and Z directions and shrinks in the
Y direction. Figure 6 shows what happens to the dimensions (Å = angstrom, one
hundred-millionth of a centimeter) of a unit cell of alpha uranium upon being heated.
As shown, heating and cooling of alpha phase uranium can lead to drastic dimensional
changes and gross distortions of the metal. Thus, pure uranium is not used as a fuel,
but only in alloys or compounds.
Figure 6 Change in Alpha Uranium Upon Heating From 0 to 300C
The beta (b) phase of uranium occurs at elevated temperatures. This phase has a
tetragonal (having four angles and four sides) lattice structure and is quite complex.
Rev. 0
Page 13
MS-01