Atomic and Nuclear Physics
DOE-HDBK-1019/1-93
RADIOACTIVITY
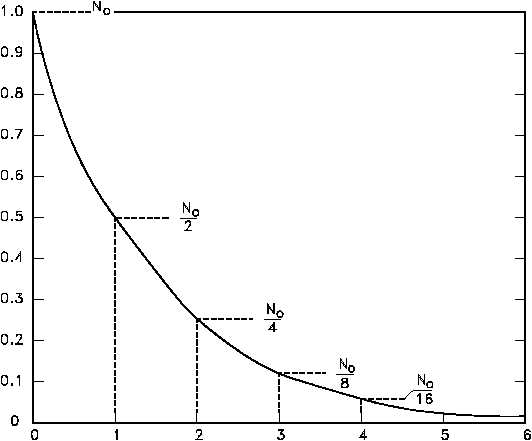
The basic features of decay of a radionuclide sample are shown by the graph in Figure 10.
Figure 10 Radioactive Decay as a Function of Time in Units of Half-Life
Assuming an initial number of atoms No, the population, and consequently, the activity may be
noted to decrease by one-half of this value in a time of one half-life. Additional decreases occur
so that whenever one half-life elapses, the number of atoms drops to one-half of what its value
was at the beginning of that time interval. After five half-lives have elapsed, only 1/32, or
3.1%, of the original number of atoms remains. After seven half-lives, only 1/128, or 0.78%,
of the atoms remains. The number of atoms existing after 5 to 7 half-lives can usually be
assumed to be negligible.
The Chemistry Fundamentals Handbook contains additional
information on calculating the number of atoms contained within a sample.
Rev. 0
Page 33
NP-01