SCINTILLATION COUNTER
Radiation Detectors
Scintillation counters are constructed by coupling a suitable scintillation phosphor to a light-
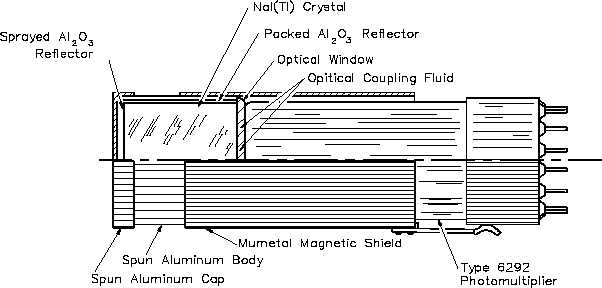
sensitive photomultiplier tube. Figure 25 illustrates an example of a scintillation counter using
a thallium-activated sodium iodide crystal.
There are three classes of solid state scintillation phosphors: organic crystals, inorganic crystals,
Figure 25 Scintillation Counter
and plastic phosphors.
Inorganic crystals include lithium iodide (LiI), sodium iodide (NaI), cesium iodide (CsI), and zinc
sulfide (ZnS). Inorganic crystals are characterized by high density, high atomic number, and
pulse decay times of approximately 1 microsecond. Thus, they exhibit high efficiency for
detection of gamma rays and are capable of handling high count rates.
Organic scintillation phosphors include naphthalene, stilbene, and anthracene. The decay time
of this type of phosphor is approximately 10 nanoseconds. This type of crystal is frequently used
in the detection of beta particles.
Plastic phosphors are made by adding scintillation chemicals to a plastic matrix. The decay
constant is the shortest of the three phosphor types, approaching 1 or 2 nanoseconds. The plastic
has a high hydrogen content; therefore, it is useful for fast neutron detectors.
IC-06
Page 46
Rev. 0